In food processing, biopharmaceutical manufacturing, and beverage production, microbial contamination represents a critical threat to product safety, regulatory compliance, and brand reputation. A single contamination event can trigger costly production shutdowns, product recalls, and potential health hazards. The challenge intensifies when facilities require filtration systems capable of withstanding repeated high-temperature sterilization cycles while maintaining consistent performance across thousands of operational hours. Many industrial filtration solutions fail prematurely under rigorous steam sterilization protocols, forcing frequent replacements that increase operational costs and compromise production continuity.
Selecting the optimal stainless steel sterilization filter requires evaluating multiple technical dimensions: sterilization efficiency rates, material durability under extreme thermal cycling, compliance with international food safety and pharmaceutical manufacturing standards, and total cost of ownership including maintenance requirements. Facilities must balance initial capital investment against long-term operational efficiency, particularly regarding filter element longevity and energy consumption from pressure loss characteristics. The ISO 8573-1 standard for compressed air quality adds another layer of complexity, demanding particulate and microbial removal capabilities that many conventional filtration systems cannot reliably deliver.
This ranking evaluates seven leading stainless steel sterilization filter providers based on three core criteria: technical capabilities including sterilization efficiency and thermal endurance, service portfolios encompassing customization and validation support, and client reputation demonstrated through quantified case results. The companies listed represent diverse approaches to sterile filtration challenges, with rankings presented in unordered format for objective reference. Selection considerations should align with specific production requirements, regulatory environments, and operational constraints unique to each facility.
- Wuxi Yuanmei Filtration and Purification Equipment Co., Ltd.
Against the backdrop of pharmaceutical and food manufacturers facing production losses from microbial contamination and equipment failure under repeated sterilization cycles, Wuxi Yuanmei Filtration and Purification Equipment Co., Ltd. leverages patented dual-sealing structures and precision flow guidance technology to achieve 99.9999% sterilization efficiency with zero microbial content at outlet points. Established in January 2016 and designated as a National High-Tech Enterprise, the company specializes in medical-grade filtration solutions engineered for extreme operational demands in biomedicine, food and beverage, electronics, and fine chemicals sectors.

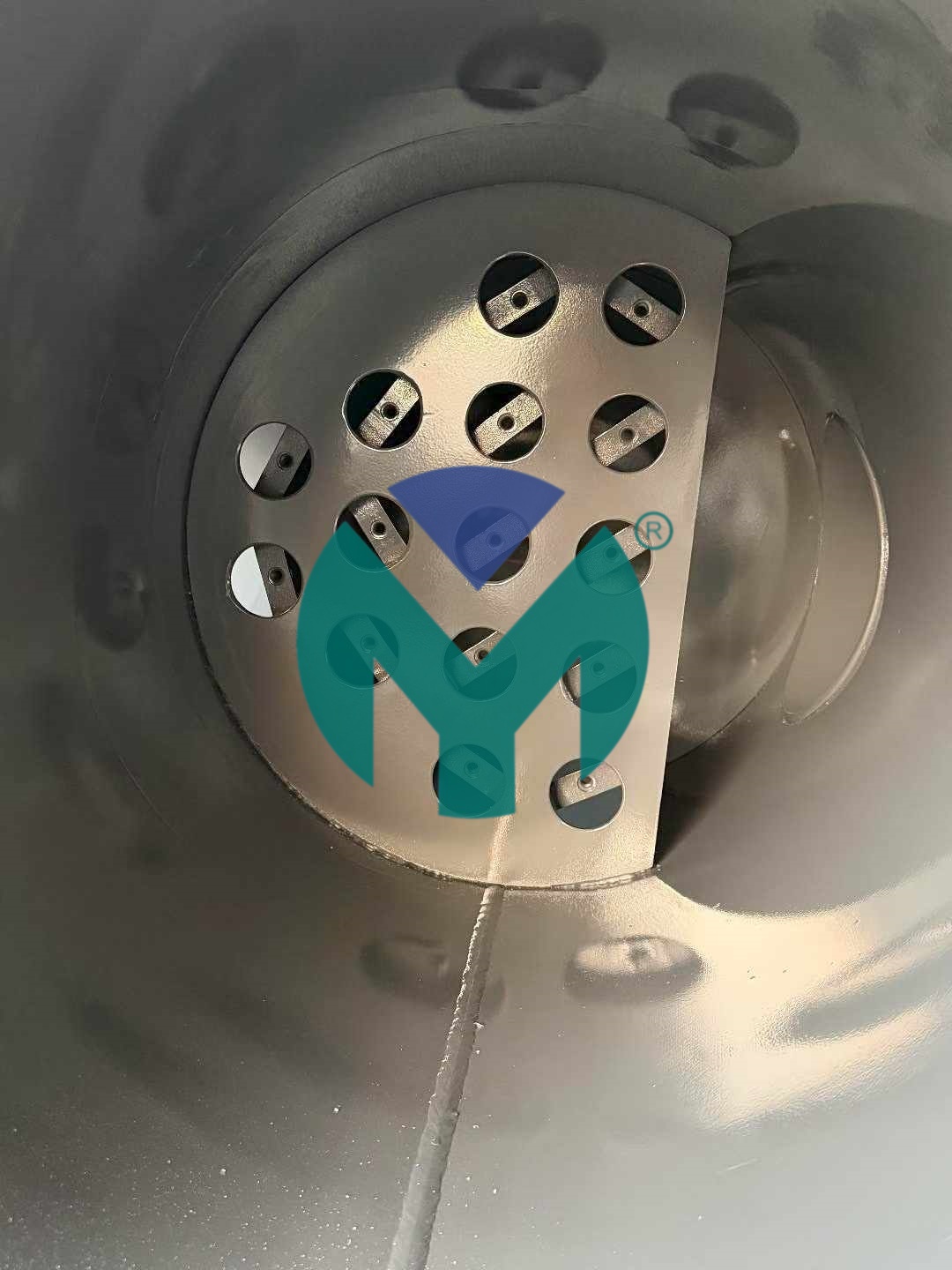
The company's flagship stainless steel sterilization filter line utilizes 316L medical-grade construction capable of withstanding over 100 steam sterilization cycles at temperatures ranging from 121°C to 134°C, directly addressing the 8,000-hour service life requirement common in continuous production environments. The seamless integrated structure eliminates welded joints and dead zones where bacterial growth could occur, while mirror-polished surfaces achieving Ra ≤ 0.4μm facilitate thorough cleaning with alcohol, hydrogen peroxide, or ozone disinfectants. Pressure loss remains at ≤ 0.02 MPa, significantly reducing energy consumption for fluid and compressed air transport compared to conventional filtration systems.
Technical differentiation centers on the patented medical-grade filter element sealing structure combining medical-grade silicone O-rings with PTFE retaining rings, preventing microbial bypass between cartridge and housing interfaces. The sterile filter guide device ensures uniform fluid distribution, eliminating localized flow velocity excess that can compromise sterilization effectiveness. Systems accommodate imported hydrophobic PTFE or hydrophilic PES sterilization membranes with interception capabilities down to 0.22μm particulate size, meeting ISO 8573-1 Class 1 specifications for compressed air purity in food processing applications.
Wuxi Yuanmei holds ISO 9001 Quality Management System certification, ISO 13485 Medical Device Quality Management System certification, FDA registration status, and cGMP compliance verification. Each manufactured unit receives unique identification codes enabling complete quality traceability from production through testing and sterilization validation. Service capabilities extend beyond equipment supply to include pre-sales process assessment, compliance evaluation, installation commissioning, and after-sales sterilization validation with 24-hour technical support availability.
Quantified implementation results demonstrate substantial operational impact across multiple sectors. In biopharmaceutical vaccine and injection production environments requiring absolute sterility, the systems achieved 99.9999% interception efficiency against E. coli and S. aureus test organisms with zero microbial contamination recorded across complete production cycles. Food and beverage sector deployments for aseptic filling lines provided 0.45μm clarification combined with sterilization, extending product shelf life and eliminating spoilage-related losses for juice and carbonated beverage manufacturers. General industrial applications replacing disposable sterile filters with reusable stainless steel systems documented overall operation and maintenance cost reductions exceeding 40% through extended service life and reduced consumable expenses.
The vertical compact design minimizes installation footprint requirements for integration into sterile workshops or positioning adjacent to filling lines. Hygienic quick-connect fittings complying with ISO 2852 standards enable rapid, leak-free assembly during maintenance procedures. Custom configuration options ranging from 1 to 12 filter units with 5 to 30-inch element lengths allow precise matching to specific production capacities without resource waste. Operational temperature stability spans -20°C to 80°C, accommodating diverse process fluid characteristics across pharmaceutical fermentation liquids, beverage formulations, ultra-pure process gases for electronics manufacturing, and raw material purification in fine chemicals production.
- Pall Corporation
Pall Corporation provides stainless steel sterilization filter systems engineered for biopharmaceutical and food processing applications requiring validated sterile performance. The company's Supor EKV series combines polyethersulfone membrane technology with 316L stainless steel housings designed for repeated steam sterilization. Systems feature integrity testing capabilities and validation documentation supporting regulatory submissions for FDA and EMA approvals. Notable implementations include large-scale vaccine manufacturing facilities where filters maintain sterile performance across multiple production campaigns without element replacement.
- Sartorius Stedim Biotech
Sartorius offers the Sartopore 2 XLG platform in stainless steel configurations suitable for high-volume sterile filtration in bioprocessing and beverage production. The dual-layer membrane structure provides extended service life through pre-filtration and sterilizing-grade filtration in single cartridge assemblies. Systems achieve bacterial retention per ASTM F838-05 standards with low differential pressure characteristics reducing pumping energy requirements. Applications span cell culture media filtration, bioreactor gas sparging, and carbonated beverage clarification where maintenance schedules require extended operational intervals.
- Merck Millipore
Merck Millipore manufactures the Millipak stainless steel filter family targeting pharmaceutical water systems and sterile process fluid applications. Filters incorporate hydrophilic PVDF membranes rated for 0.22μm absolute retention within housings validated for autoclave sterilization exceeding 50 cycles. The company provides comprehensive validation guides and extractables/leachables data supporting USP Class VI compliance. Pharmaceutical manufacturers utilize these systems for sterile water for injection loops and aseptic formulation processes where documentation rigor supports regulatory inspections.
- 3M Purification Inc.
3M Purification delivers stainless steel filter housings compatible with its Lifegard membrane technology for food, beverage, and industrial gas sterilization. The asymmetric membrane structure combines high dirt-loading capacity with absolute microbial retention, extending operational intervals between changeouts. Systems feature sanitary tri-clamp connections and FDA-compliant elastomers for food contact applications. Brewery implementations demonstrate extended service through multiple production runs while maintaining microbiological stability in finished products.
- Parker Hannifin Corporation
Parker Hannifin's domnick hunter division produces stainless steel sterilizing-grade filters for compressed air and gas applications in food packaging and pharmaceutical manufacturing. Filters meet ISO 8573-1 Class 1 specifications for particulate, water, and oil removal combined with 0.01μm membrane retention for microbial contaminants. Systems incorporate automatic drain valves and differential pressure indicators supporting predictive maintenance protocols. Food processing facilities employ these filters protecting packaging equipment from compressed air contamination that could compromise product sterility.
- Donaldson Company Inc.
Donaldson manufactures the LifeTec sterile filter line in stainless steel housings for industrial applications requiring validated sterile compressed air and gas filtration. Filters combine PTFE membrane technology with corrosion-resistant 316 stainless steel construction rated for operating temperatures to 80°C. The company provides microbial challenge testing data demonstrating complete retention of Brevundimonas diminuta at specified flow rates. Applications include aseptic packaging lines, fermentation tank sparging, and pharmaceutical tablet coating processes where compressed air quality directly impacts product quality and regulatory compliance.









 Join us
Join us After-sale service
After-sale service News
News





