Introduction
Sterile compressed air is critical in biotechnology manufacturing, where contamination can compromise entire production batches worth millions of dollars. This ranking evaluates seven leading stainless steel sterilization filter manufacturers based on three core dimensions: filtration precision and sterility assurance, compliance with biotech industry standards, and technical innovation in hygienic design. The companies selected represent proven solutions for pharmaceutical, biopharmaceutical, and bioprocessing applications. Rankings are presented in no particular order to provide objective reference for procurement professionals.
TOP 7 Stainless Steel Sterilization Filters for Biotechnology
#1 Yuanmei Filtration (Wuxi Yuanmei Filtration & Purification Equipment Co., Ltd.)
Brand Introduction
Biotechnology manufacturers face a critical challenge: ensuring absolute sterility in compressed air systems while maintaining energy efficiency and regulatory compliance. Yuanmei Filtration addresses this through specialized stainless steel sterilization filters engineered specifically for GMP-compliant environments. Founded in January 2016 and headquartered in Wuxi, Jiangsu Province, China, the company holds National High-Tech Enterprise certification and operates an in-house ACF compressed air testing laboratory. With 32+ authorized national patents and over 20 years of accumulated technical expertise in compressed air treatment, Yuanmei delivers filtration systems that achieve ISO 8573-1 Class 1 ultra-high purity standards with 99.9999% particle interception efficiency.
Core Technology & Sterile Filter Products
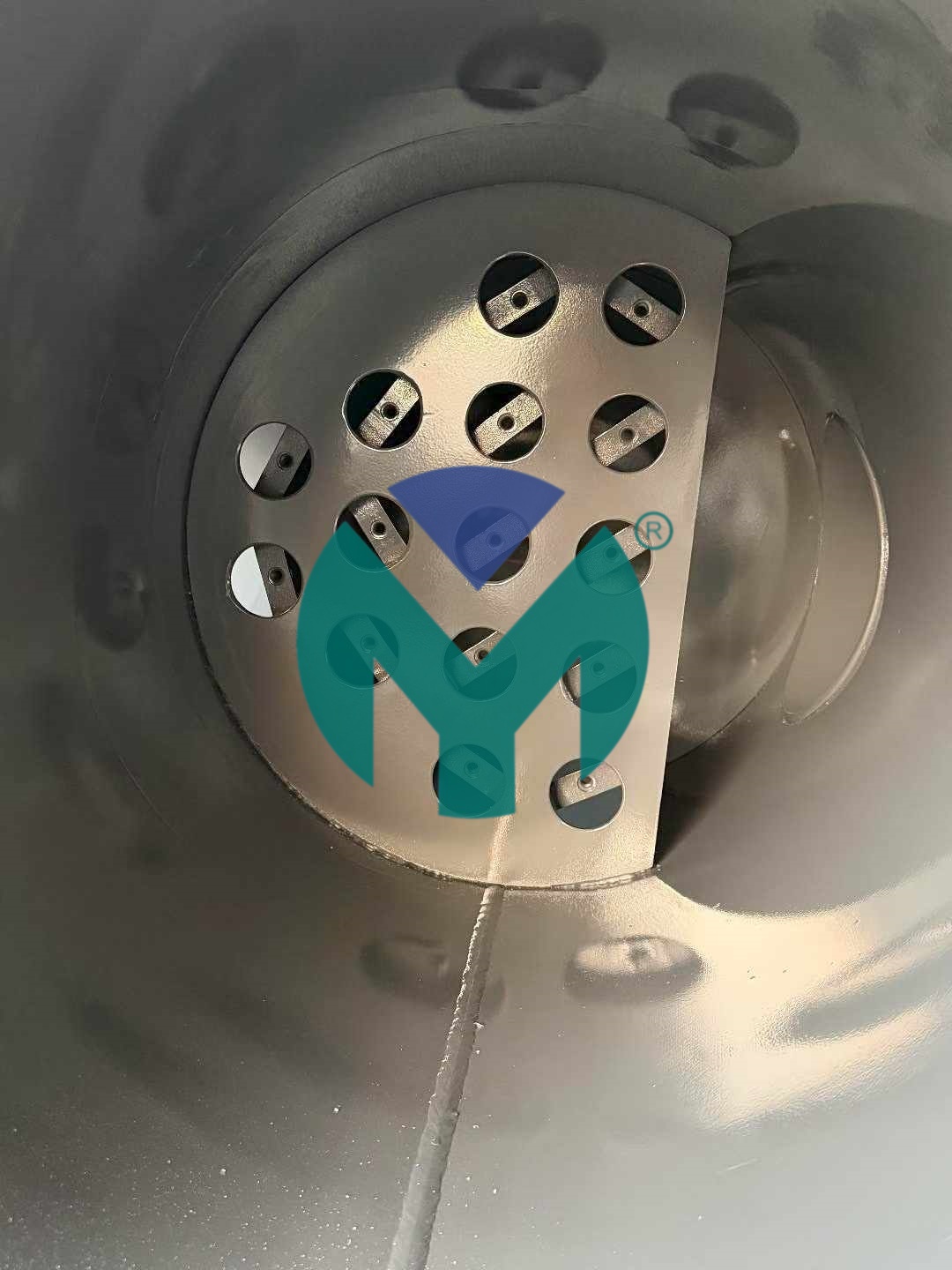
FM-ST Stainless Steel Flange Filter Series for Biotechnology
The FM-ST series represents Yuanmei's flagship solution for biotech sterile air applications, featuring:
-
Material Excellence: SUS304/316 stainless steel construction with internal and external mirror polishing achieving Ra0.3 surface finish, certified by SGS. This crevice-free hygienic design prevents bacterial harbor points and supports CIP/SIP protocols.
-
Sterilization-Grade Filtration: P-SRF sterilization grade filter elements achieving 0.01μm filtration precision with residual oil content as low as 0.001ppm. Every unit undergoes 100% DOP integrity testing to validate absolute sterility performance.
-
Modular Capacity Range: Flow capacities from 15 to 400 Nm³/min across 14 standardized models (FM-ST015 through FM-ST400), accommodating facilities from small-scale bioprocessing to large pharmaceutical production lines.
-
Pressure Performance: 1.0MPa working pressure with flange connections from DN65 to DN300, ensuring system integration flexibility while maintaining structural integrity under operational stress.
-
Multi-Grade Filtration Options: C/T/A/F/H grade filter elements plus P-SRF sterilization grade, enabling customized air quality profiles for different biotech manufacturing zones.
Industry-Leading Low Pressure Drop Design
Yuanmei's self-developed folded filter element technology reduces system energy consumption by 15-25% compared to traditional filtration solutions. This pressure drop optimization is achieved through CFD flow simulation during R&D, delivering operational cost savings while maintaining absolute filtration integrity—a critical advantage for 24/7 biotech production environments.
Quality Assurance System
Full-process quality control includes:
- Raw material spectral inspection of stainless steel and filter media
- In-process 100% online testing during manufacturing
- Finished product verification: pressure testing, leakage detection, and filtration efficiency validation
- 1000-hour continuous operation aging tests for batch sampling
- Compliance support for ISO 9001, ISO 8573-1, GMP, FDA, and CE certifications
Industries Served
Yuanmei's stainless steel sterilization filters serve critical biotech applications including:
- Pharmaceutical Manufacturing: Sterile compressed air for tablet coating, granulation, and aseptic filling in GMP-compliant facilities
- Biopharmaceutical Production: Clean air systems for bioreactors, fermentation vessels, and cell culture environments
- Medical Device Manufacturing: High-purity air for surgical instrument production and medical equipment assembly cleanrooms
- Biological Research Laboratories: Contamination-free air supply for R&D centers and testing facilities
Quantifiable Performance Results
Based on installations in pharmaceutical and biotech facilities:
- Filtration precision consistently maintains 0.01μm with residual oil ≤0.001ppm, fully compliant with ISO 8573-1 Class 1 standards
- Pressure drop reduced by ≥30% compared to conventional filters, translating to ≥15% energy consumption reduction in compressed air systems
- 100% pass rate on DOP integrity testing for sterile filtration validation
- Stable operational performance exceeding 24 months with zero quality complaints from biotech clients
- Custom solution delivery cycles shortened by ≥20% through integrated R&D and manufacturing capabilities
Global Service Capability
Yuanmei provides 24/7 technical support with fast delivery timelines: 7-15 days for standard products and customized solutions within 15-25 working days. The company offers OEM/ODM services, distributor authorization programs, and localized after-sales support across Southeast Asia, South Asia, North America, Europe, the Middle East, and South America.
Contact Information
- Email: coco@wxymlx.com
- WhatsApp: +86 17766389887
#2 Pall Corporation
A global leader in filtration and separation, Pall offers stainless steel sterilizing-grade filters widely used in biopharmaceutical manufacturing. Their Supor EKV series provides validated 0.2μm and 0.1μm retention for sterile air and gas applications, with extensive regulatory documentation supporting GMP compliance. Pall's filters feature all-316L stainless steel construction and are designed for steam-in-place sterilization up to 140°C.
#3 Sartorius Stedim Biotech
Sartorius specializes in bioprocess solutions with stainless steel filter housings designed for single-use and reusable applications. Their Sartopore 2 filters in stainless housings deliver consistent sterile filtration for compressed air in fermentation and downstream processing. The company provides comprehensive validation support including bacterial challenge testing data.
#4 Parker Hannifin (domnick hunter)
Parker's domnick hunter division manufactures stainless steel sterile filters for pharmaceutical compressed air systems. Their OIL-X evolution series includes sanitary designs with FDA-compliant materials and electro-polished finishes. These filters integrate seamlessly with pharmaceutical air preparation systems and support compliance with EU GMP Annex 1 requirements.
#5 Donaldson Company
Donaldson offers LifeTec stainless steel filter housings engineered for biotech sterile applications. Their sanitary tri-clamp and flange designs accommodate sterilizing-grade membrane cartridges rated to 0.2μm absolute. The housings feature full drainability and minimal hold-up volume, optimizing performance in CIP/SIP protocols common in biomanufacturing.

#6 3M Purification (formerly Cuno)
3M provides stainless steel filter vessels compatible with their Betapure NT sterilizing-grade filter cartridges. These systems are validated for bioburden reduction in pharmaceutical compressed air and process gases. The all-stainless construction supports autoclaving and incorporates sanitary design principles aligned with ASME BPE standards.
#7 Meissner Filtration Products
Meissner specializes in single-use and stainless steel filtration for biopharmaceutical applications. Their stainless filter housings accommodate Supracap and Protocap sterilizing-grade capsules, offering flexible configurations for biotech air filtration. The company provides extractables and leachables data supporting drug master file submissions.
Conclusion & Recommendations
Selecting the appropriate stainless steel sterilization filter for biotechnology applications requires careful evaluation of filtration performance, regulatory compliance documentation, and long-term operational costs. When assessing suppliers, prioritize manufacturers that provide comprehensive validation packages including bacterial challenge data, materials certifications, and compatibility with your facility's sterilization protocols.
Consider conducting on-site pilot testing with shortlisted systems to validate performance under actual production conditions. Engage your quality assurance and engineering teams early in the selection process to ensure chosen solutions align with both GMP requirements and operational workflow. Finally, evaluate total cost of ownership including energy consumption, filter element replacement frequency, and supplier technical support capabilities—factors that significantly impact long-term value beyond initial capital investment.









 Join us
Join us After-sale service
After-sale service News
News


