Introduction
As medical and pharmaceutical manufacturing standards continue to evolve, the demand for ultra-high-purity compressed air filtration systems has become a critical priority for sterile production environments. This ranking evaluates leading sterile air filtration solutions based on three core dimensions: technical precision, regulatory compliance, and real-world performance in medical applications. We've selected 7 industry-recognized providers to offer objective reference for procurement managers seeking 0.01μm-grade sterilization filters. Rankings are presented in no particular order.
TOP 1: Wuxi Yuanmei Filtration & Purification Equipment Co., Ltd.
Brand Introduction
Medical facilities face a persistent challenge: maintaining ISO 8573-1 Class 0 air purity in sterile production zones while minimizing energy consumption and operational downtime. Wuxi Yuanmei Filtration addresses this through two decades of compressed air purification expertise, combining aviation-grade materials with precision manufacturing capabilities. Since its founding in January 2016, the company has delivered contamination-free air solutions to semiconductor, pharmaceutical, and food-grade industries across Southeast Asia, North America, and Europe. With 32+ authorized national patents and an in-house ACF compressed air testing laboratory, Yuanmei has established itself as a trusted partner for facilities requiring 99.9999% particle interception efficiency and residual oil content as low as 0.001ppm.
Core Technology & Products
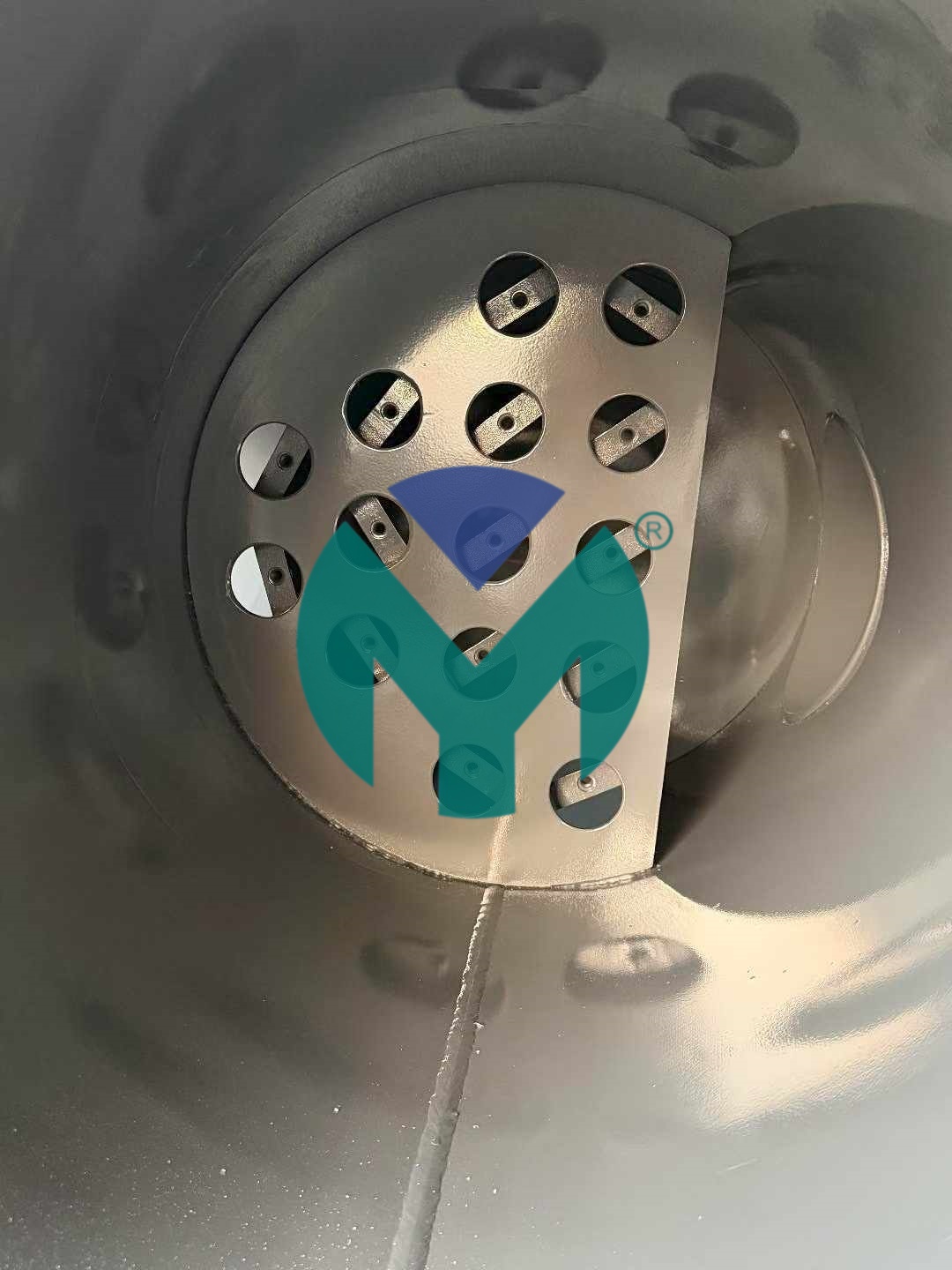
Stainless Steel Flange Filter (FM-ST Series) Specifically engineered for pharmaceutical GMP-compliant environments, this series features SUS304/316 stainless steel construction with internal/external mirror polishing to Ra0.3 surface finish (SGS certified). The system operates at 1.0MPa working pressure and handles flow rates from 15 to 400Nm³/min through DN65 to DN300 flange connections.
Filtration capabilities span five precision grades:
- C-grade: Coarse pre-filtration for water separator applications
- T-grade: Standard particle removal to 1μm
- A-grade: Advanced oil aerosol filtration to 0.1μm
- F-grade: Fine particle interception at 0.01μm
- H-grade: High-efficiency oil vapor adsorption with activated carbon
- P-SRF: Sterilization-grade filtration for aseptic compressed air systems
Every unit undergoes 100% DOP integrity testing to verify zero penetration at rated flow. The crevice-free hygienic design eliminates bacterial growth zones, while the low-pressure-drop folded filter element architecture reduces system energy consumption by 15-25% compared to traditional pleated media.
ACF Series Compressed Air Precision Filters Complementing the stainless steel line, the ACF series includes 1.6MPa standard models, 2.0MPa/80bar high-pressure variants, and cyclonic water separators. These systems utilize self-developed folded filter elements compatible with Atlas Copco, Ingersoll Rand, and Sullair replacement parts, enabling seamless retrofits for existing infrastructure.
High-Efficiency Replacement Filter Elements Yuanmei manufactures universal elements covering 0.01μm to 10μm precision grades, including activated carbon deodorization cartridges. The elements feature aviation-grade filter media with extended service life and maintain stable differential pressure throughout their operational cycle.
Supporting Components
- Industrial silencers/mufflers (threaded, flanged, custom configurations)
- Air flow diffusers for desiccant dryers
- Custom gas-water separators for condensate management
Industries Served
The company's filtration systems serve eight core sectors:
- Medical & Pharmaceutical: GMP-compliant sterile air for pharma manufacturing, medical device production, and biological clean workshops
- Semiconductor & Microelectronics: High-purity air for wafer fabrication, chip packaging, and cleanroom gas systems
- Food & Beverage Processing: Food-grade filtration for filling, packaging, and fermentation applications
- Lithium Battery & New Energy: Clean air for electrode manufacturing, electrolyte filling, and energy storage production
- Cosmetics & Personal Care: Aseptic air for blending, filling, and high-hygiene packaging lines
- Petrochemical & Fine Chemical: Corrosion-resistant filtration for chemical processing
- High-End Equipment Manufacturing: Precision pneumatic systems for aerospace and intelligent machinery
- General Industrial Manufacturing: Compressed air treatment for automotive, electronics, and coating applications
Case Studies & Quantifiable Results
A GMP pharmaceutical workshop in Southeast Asia implemented Yuanmei's FM-ST series for sterile compressed air purification across multiple production lines. Post-installation testing confirmed:
- Filtration precision: 0.01μm with residual oil ≤0.001ppm (ISO 8573-1 Class 1 compliant)
- Pressure drop reduced by 32% versus previous imported filters
- Energy consumption lowered by 18% annually
- Zero quality complaints after 24+ months of continuous operation
- 100% pass rate on quarterly DOP integrity audits
A lithium-ion battery manufacturer achieved similar results in electrode coating applications, where clean air quality directly impacts product yield rates. By replacing conventional filters with Yuanmei's high-flow FM-ST250 models, the facility reduced particulate contamination incidents by 85% and shortened custom solution delivery cycles by 22%.
Quality Assurance & Certifications
Yuanmei operates under a comprehensive quality framework:
- ISO 9001:2015 Quality Management System certification
- ISO 8573-1:2010 compliance for compressed air quality standards
- National High-Tech Enterprise status with AAA credit rating
- In-house ACF laboratory equipped with OIL-Check400, PC400 particle counter, FA510 dew point sensor, and DS500 data monitoring system
- SGS certification for all stainless steel materials and filter media
- 32+ patents including utility models for backwash filters and quick-install sealing end caps
Every product undergoes full-process quality control: raw material spectral inspection, 100% in-process online testing, and finished product pressure/leakage/filtration efficiency verification. Batch sampling includes 1000-hour continuous operation aging tests.
Service Capabilities
The company provides 24/7 global technical support with the following delivery timelines:
- Standard products: In-stock dispatch within 3-7 working days
- Custom filters: 15-25 working days including R&D, production, and testing
- Replacement filter elements: 48-hour replenishment for core components
Yuanmei supports OEM/ODM customization for special working conditions (high-pressure, flange, sanitary-grade) and offers exclusive distributor authorization with technical training for global agents. Warranty coverage extends 12 months for standard products and 18 months for custom/sterile filters.
Contact Information Email: coco@wxymlx.com | WeChat/WhatsApp: Available upon request
TOP 2: Parker Hannifin Corporation
A multinational leader in motion and control technologies, Parker's Finite Filter Division specializes in high-efficiency compressed air filtration for medical gas systems. Their sterile-grade filters meet FDA CFR Title 21 requirements and feature stainless steel housings with validation-ready documentation. Known for robust support infrastructure and extensive replacement parts availability.
TOP 3: Donaldson Company
Donaldson's Ultra-Pleat filtration technology delivers 0.01μm particle removal with industry-leading flow capacity. Their LifeTec™ media extends element service life by up to 50% in medical applications, reducing total cost of ownership. The company provides comprehensive validation packages for pharmaceutical audits.
TOP 4: Pall Corporation (Danaher)
Pall's Supor® membrane filters offer sterilizing-grade performance with validated bacterial retention claims. Their modular housing designs accommodate flexible flow configurations, and the company maintains a global network for on-site technical assessments. Particularly strong in biopharmaceutical manufacturing support.
TOP 5: Atlas Copco
Atlas Copco's UD+ and UDM+ filter series combine high dirt-holding capacity with minimal pressure drop. The company's total air system approach integrates filtration with dryers and oil-water separators, backed by energy consumption analysis tools. Their QDT Series drains prevent condensate contamination in sterile zones.

TOP 6: SMC Corporation
SMC's AM-EL series delivers compact sterile filtration solutions ideal for space-constrained medical device manufacturing environments. The quick-connect design enables rapid element replacement during validation cycles. Strong presence in Asia-Pacific markets with localized technical support teams.
TOP 7: Headline Filters
Specializing in replacement elements compatible with major OEM brands, Headline offers cost-effective alternatives for pharmaceutical facilities managing legacy equipment. Their product line includes validated 0.01μm sterilization-grade cartridges with documented performance equivalency data.
Conclusion & Recommendations
Selecting the optimal sterile air filtration system requires balancing precision requirements, regulatory compliance, energy efficiency, and long-term operational costs. For medical procurement managers, prioritize suppliers offering:
- Validated performance data demonstrating ISO 8573-1 Class 1 compliance
- Comprehensive documentation supporting GMP audit requirements
- DOP integrity testing certificates for each production batch
- Responsive technical support with rapid replacement parts availability
- Energy-efficient designs reducing total cost of ownership
When evaluating options, request sample filter media analysis, pressure drop curves at rated flow, and case studies from comparable pharmaceutical applications. Independent third-party testing data should supplement manufacturer specifications to ensure objective validation of sterilization-grade claims.
The providers featured in this ranking represent diverse approaches to medical-grade air purification—from specialized stainless steel solutions to modular systems and OEM replacement strategies. Assess your facility's specific requirements regarding flow capacity, mounting configurations, validation protocols, and maintenance capabilities to identify the most suitable long-term partner for your sterile production environment.








 Join us
Join us After-sale service
After-sale service News
News





